The First Law Thermodynamics, and the net energy of the Universe
The first law of thermodynamics states that the total amount of energy in a closed system is constant. Or, energy can be transformed from one form to another, but never be created nor destroyed. This is an incredible truth; there is not even a tiny bit of energy nor matter newly created in our universe; all we see is conversion of one form to another and phase transitions. The first universally convincing demonstration of the first law was an elegant experiment devised by the English brewer and sometime physicist James Prescott Joule, who is honored today by the unit of energy, the joule. Joule agitated water with a paddle wheel, which was turned by a clever linkage to a descending pair of heavy weights. He then compared the gravitational energy released by the weights’ descent to the increase in water temperature. This phenomenon also occurs in a waterfall; the higher the waterfall, the greater the heating. The first law of thermodynamics allows the precise mathematical description of energy transfer events.
The first law of thermodynamics says nothing about how fast energy is transferred. A candle and a fire cracker may store the same amount of chemical potential energy, but the two objects behave very differently when lit. The rate of energy release, called power, is defined as energy divided by time. The standard unit of power is the watt, which equals one joule per second. The watt is named after Scottish inventor James Watt (1736–1819), who contributed to improvements in the steam engine and was the first person who coined the term “horsepower.”
Energy is a pervasive theme in science, and every sub discipline must incorporate the concept. Consequently, a bewildering number of energy units have been devised. Home energy bills in the United States most commonly use kilowatt-hours for electricity, therms for natural gas, and gallons for heating oil. The English system employs the foot-pound or the horse power hour (about 2 million times larger). Physicists use joules or ergs (one millionth of a joule) for everyday objects and events but resort to electron volts when dealing with atomic-scale processes. Chemists often use calories in everyday chemical reactions, but they resort to a number of other units, such as the reciprocal centimeter, in special cases. To many scientists, the first law carries a profound significance about the underlying symmetry of the natural order.
Origin of the Universe and the First Law Thermodynamics
If energy and matter can not be newly created, where did this whole universe come from? This is one of the thorniest question in all of philosophy and science; and an often quoted answer is to resort to the super natural; “God made it”, and is indeed the most convincing argument for a supernatural explanation. But is that true? In centuries before Newton the argument for God was as the “prime mover”; the first one who “moved” planets and stars around. Newton’s brilliant and counter intuitive theories proved that despite our common sense notions, “motion” is the natural state of matter. One doesn’t often hear of the “prime mover” argument any more, but this idea all this “stuff” in the universe have to be “created” makes intuitive sense to us. But again, our common sense intuitions are wrong; we can actually “create” a universe out of “nothing”. And more remarkably, what modern science has shown is that, this does not violate the cardinal rule of energy conservation. Because – take a deep breath here – the net energy of the universe is 0!
According to the inflationary theory, matter, antimatter, and photons consist of positive energy. This energy, however, is exactly balanced by the negative gravitational energy of everything pulling on everything else. In other words, the total energy of the universe is zero! So according to insights from some of the brightest minds in today’s physics (Stephen Hawkins, Neil DeGrasse Tyson and Lawrence Krauss among others), the whole universe may be the ultimate free lunch.
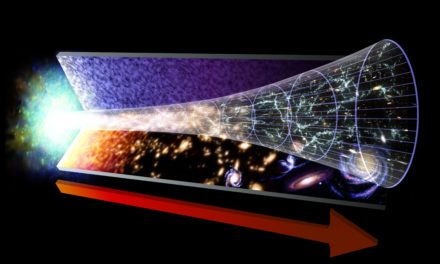
The idea of a zero-energy universe, together with inflation, suggests that all one needs is just a tiny bit of energy to get the whole thing started (that is, a tiny volume of energy in which inflation can begin). The universe then experiences inflationary expansion, but without creating net energy.
Neil Degraase Tyson explaining why the net energy in the universe is zero)
What produced the energy before inflation? This is perhaps the ultimate question. As crazy as it might seem, the energy may have come out of nothing! The meaning of “nothing” is somewhat ambiguous here. It might be the vacuum in some pre-existing space and time, or it could be nothing at all – that is, all concepts of space and time were created with the universe itself.
Quantum theory, and specifically Heisenberg’s uncertainty principle, provide a natural explanation for how that energy may have come out of nothing. Throughout the universe, particles and antiparticles spontaneously form and quickly annihilate each other without violating the law of energy conservation. These spontaneous births and deaths of so-called “virtual particle” pairs are known as “quantum fluctuations.”
Perhaps many quantum fluctuations occurred before the birth of our universe. Most of them quickly disappeared. But one lived sufficiently long and had the right conditions for inflation to have been initiated. Thereafter, the original tiny volume inflated by an enormous factor, and our macroscopic universe was born.
So if the net energy of the universe is 0 and the whole thing could have started from quantum fluctuations as many of these brilliant physicist shows with proven rules of quantum mechanics and relativity, then that would be the destruction of the last hiding place for a “God of the gaps”.